How to Remove Sulfur from Used Oil?
Written By: Mr.Ran
Senior Petrochemical & Waste Oil Recycling Engineer
Deeply involved in the design, manufacturing, and optimization of various waste oil recycling and petrochemical equipment, delivering practical and efficient solutions for clients worldwide.
Sulfur is a hard limit in used oil refining. If the amount is too high, the grade of base oil is automatically downgraded – or even fails to meet specifications. European standard V demands diesel with less than 10 ppm of sulfur content. For group II base oil, it is 300 ppm. And for group III base oil, it is less than 100 ppm.
In this blog, we will discuss three industrial processes for sulfur removal from used oil: Hydrodesulfurization, Oxidative Desulfurization, and the Adsorption process.
Hydrodesulfurization (HDS) — The Industrial Standard
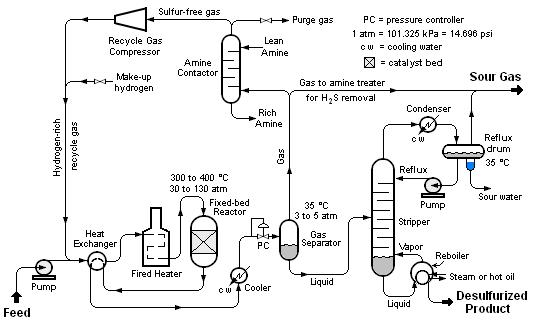
Desulfurization by HDS is done through the use of hydrogen to treat oils at elevated temperatures and pressures. Common conditions: 200°C to 300°C, 3.0MPa to 4.0MPa. Organic sulfur is made into H₂S using NiMo or CoMo catalysts, which is subsequently stripped out.
The problem with used oil: it contains silicon (Si) from antifoam agents and boron (B) from additives. Both poison catalysts.
Two-reactor vs. three-reactor
| Configuration | Desulfurization (week 5) | Catalyst life |
| Two-reactor | ~85% | ~4 weeks |
| Three-reactor (with guard bed) | >97% | ~2x longer |
The three-reactor configuration includes an independent guard bed to capture Si and B before reaching the main reactor. This is the recommended design for units > 50 tons/day requiring Group II/III or Euro V diesel.
Considerations:
- Very high capital cost (pressure vessels, hydrogen compressor)
- Consumption of hydrogen continuously
- Requires H₂S recovery downstream (see Section 4)
Oxidative Desulfurization (ODS) — A Hydrogen-Free Alternative
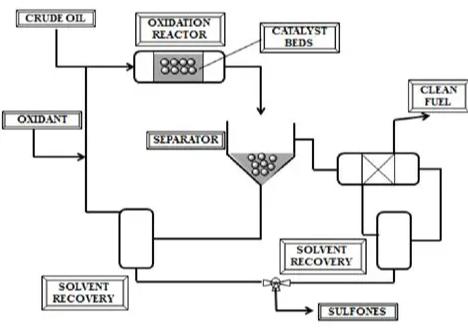
In ODS, a chemical reaction takes place without pressure. An oxidant, usually hydrogen peroxide along with phosphotungstic acid catalyst, converts sulfides and thiophenes into sulfones. The latter compound is polar in nature and easily separates from the oil through solvents.
Reaction Conditions: Temperature at 90°C, atmospheric pressure, reaction time of 15-60 minutes.
Reported efficiency for used oil: 85–92%, depending on feedstock and oxidizer dosage.
Where Does ODS Fit?
- Small-scale refineries (capacity <50 tons/hour)
- Targeting Group I base oils/industrial fuel oil feedstocks
- Less initial investment compared to HDS
Drawbacks:
- ODS cannot achieve Euro V levels on its own (<10 ppm)
- Generates sulfones in waste streams
- High chemical requirements per ton of crude
Adsorptive Desulfurization — A Polishing or Standalone Option
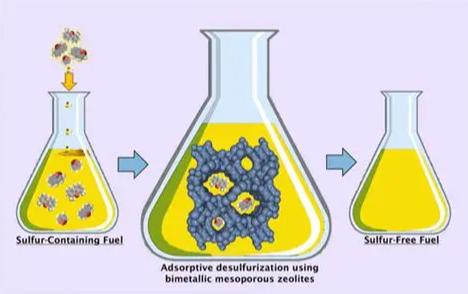
Adsorption captures sulfur compounds on porous materials. Activated carbon, alumina, and clay all work. Efficiency varies significantly by adsorbent and bed configuration.
Laboratory data (activated carbon):
| Bed configuration | Sulfur removal |
| Single bed | ~28% |
| Three beds in series | ~92.5% |
As a standalone process, adsorption is practical only below 20 tons/day. Above that, frequent adsorbent replacement or regeneration drives operating costs up.
As a polishing step, adsorption works well after HDS or ODS. It pulls sulfur from hundreds of ppm down to fifty or lower. Many Group II refineries use a clay or carbon guard bed as their final step.
H₂S Management — A Required Downstream Unit
For any HDS process, the sulfur comes out of the oil as H₂S gas when you use HDS. H₂S is toxic. It’s regulated very tightly by governments.
Two proven routes for used oil refineries:
| Scale | Technology | Outlet H₂S |
| Large (>100 t/d) | Amine + Claus sulfur recovery | <10 mg/m³ |
| Small to medium | Chelated iron (e.g., GLT) | <1 mg/m³ |
Chelated-iron methods are less expensive than Claus recovery systems for smaller HDS facilities using used oils (<30 t/d sulfur produced). This is supported by several practical uses in refineries on tail gases and off-gases from hydrotreaters.
Remember: Allocate funding for H₂S removal before designing an HDS facility. It’s integral to the process.
How to Select?
Three variables determine the right desulfurization method: scale, target product, and capital available.
| Scale | Target Product | Recommended Method |
| ≥50 tons/day | Group II/III base oil or Euro V diesel | Three-reactor HDS + H₂S recovery |
| 20–50 tons/day | Group I base oil or industrial fuel | ODS + adsorption polishing |
| <20 tons/day | Pretreatment or fuel blending | Adsorption only |
| Any scale | H₂S tail gas | Amine/Claus or chelated iron |
For new builds targeting high-value base oils, HDS is more economical over the full facility lifecycle despite higher upfront cost. For retrofits or smaller operators, ODS offers a lower entry point.
Purepath Ultrasonic oxidative desulfurization (UODS)

PurePath’s ultrasonic oxidative desulfurization plant uses mechanical ultrasound waves to intensify the oxidation reaction. The result is faster reaction kinetics and lower chemical consumption compared to conventional ODS.
Key specifications from operating plants:
| Parameter | Typical Value |
| Outlet sulfur | <10 ppm |
| Oil yield | 90–99% |
| Chemical cost | US$6–35 per ton of feed |
| Manpower | 2–10 operators |
| Lead time | 90–120 days |
The plant removes not only sulfur but also nitrogen compounds, gums, and asphaltenes. It handles diesel, naphtha, kerosene, pyrolysis oil, and base oil as feedstock. By-product sulfones can be sold as fuel oil.
The unit is available as a standalone desulfurization plant or as part of a larger refinery ecosystem — PurePath also supplies waste oil recycling and lube oil refining plants that integrate with the UODS for a complete, environmentally compliant setup.







